of Victoria
SciencesNavigation

BA (Man), MSc (Calgary), PhD (Calgary)
Synaptic plasticity, stroke, diabetes, in vivo imaging
Dr. Brown's neurobiology lab employs in vivo microscopic imaging technologies such as two-photon microscopy and functional imaging of neuronal and hemodynamic activity that allow the visualization of neuronal structures deep within the living brain or the processing of sensory information in real-time.
A central goal of Dr. Brown's research program is to use these experimental approaches to characterize the neurobiological mechanisms that allow the cerebral cortex to develop normally and change throughout life in response to new experiences (eg. learning, drug exposure) or pathology such as stroke or diabetes.
Donations support our students and faculty in their mission to produce internationally recognized advances in neuroscience knowledge.
Support Dr. Craig Brown's research.
The Brown lab is actively looking for talented new trainees at the graduate and post-doctoral level. Please contact Dr. Brown (brownc@uvic.ca) with your CV including reference list, unofficial transcripts and a research statement.
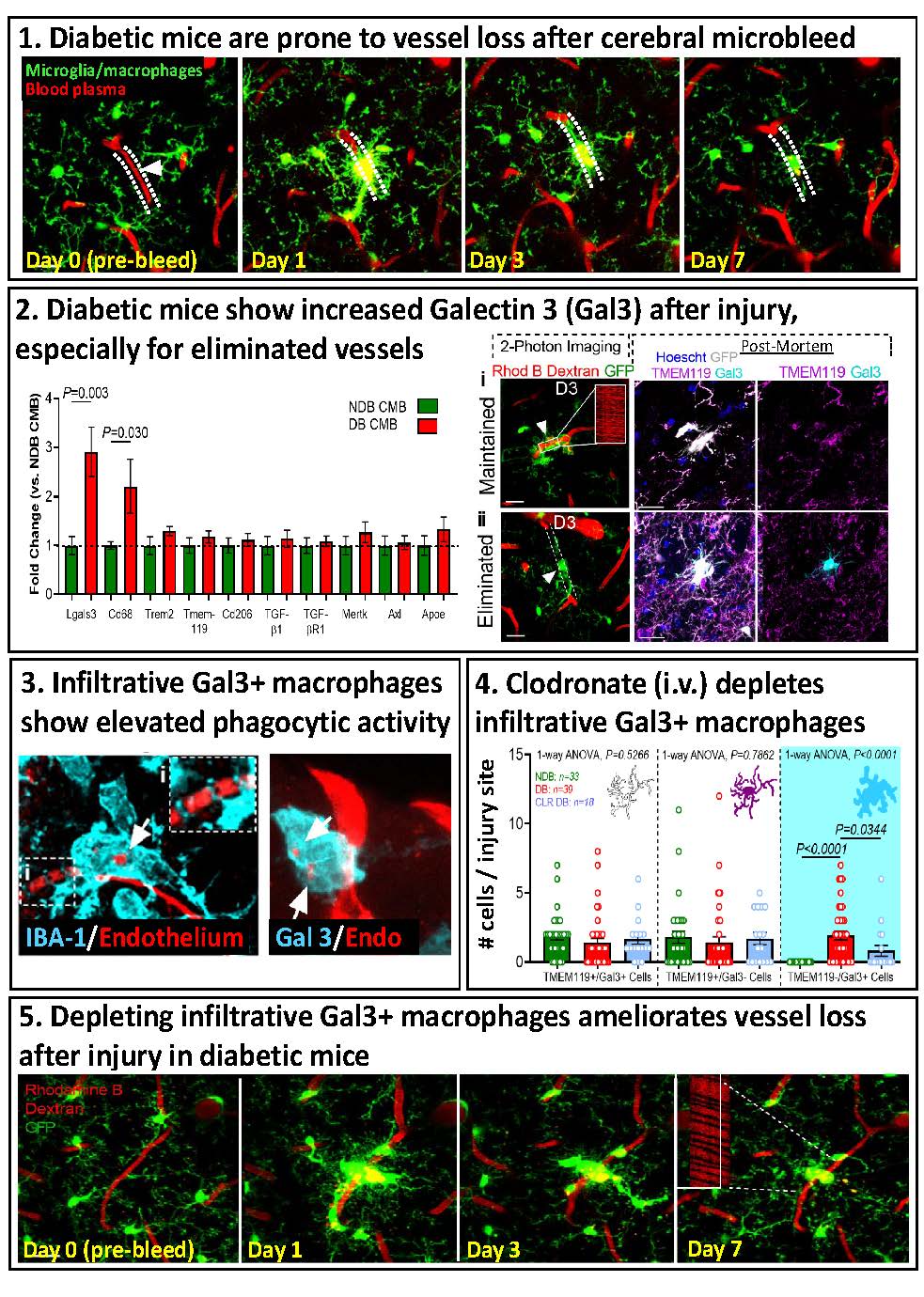
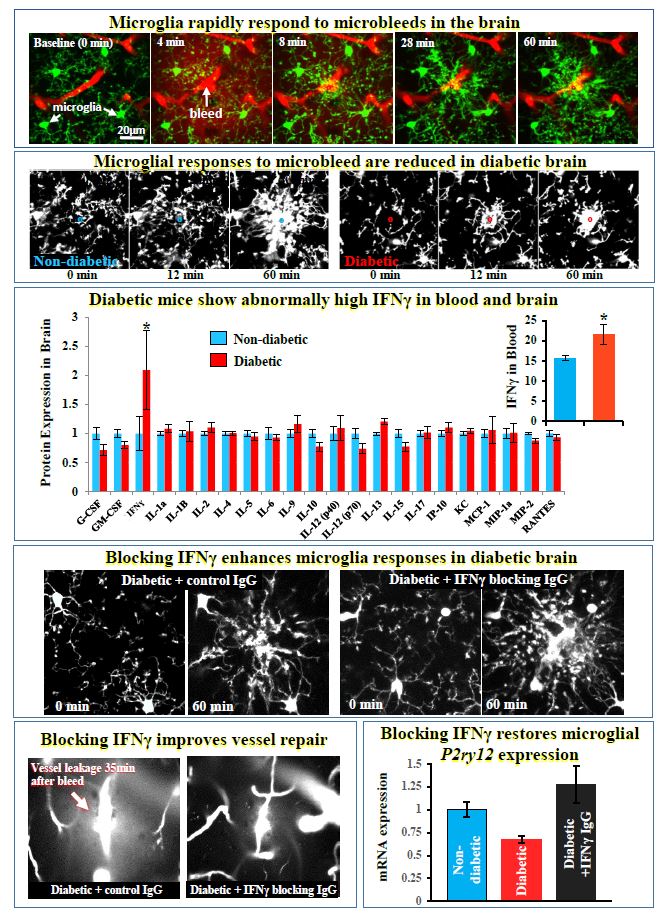
For more information: Taylor S, Mehina E, White E, Reeson P, Yongblah K, Doyle KP, Brown CE. 2018. Suppressing interferon gamma stimulates microglial responses and repair of microbleeds in the diabetic brain. Journal of Neuroscience Sep 12. pii: 0734-18. doi: 10.1523/JNEUROSCI.0734-18.2018. [Epub ahead of print]
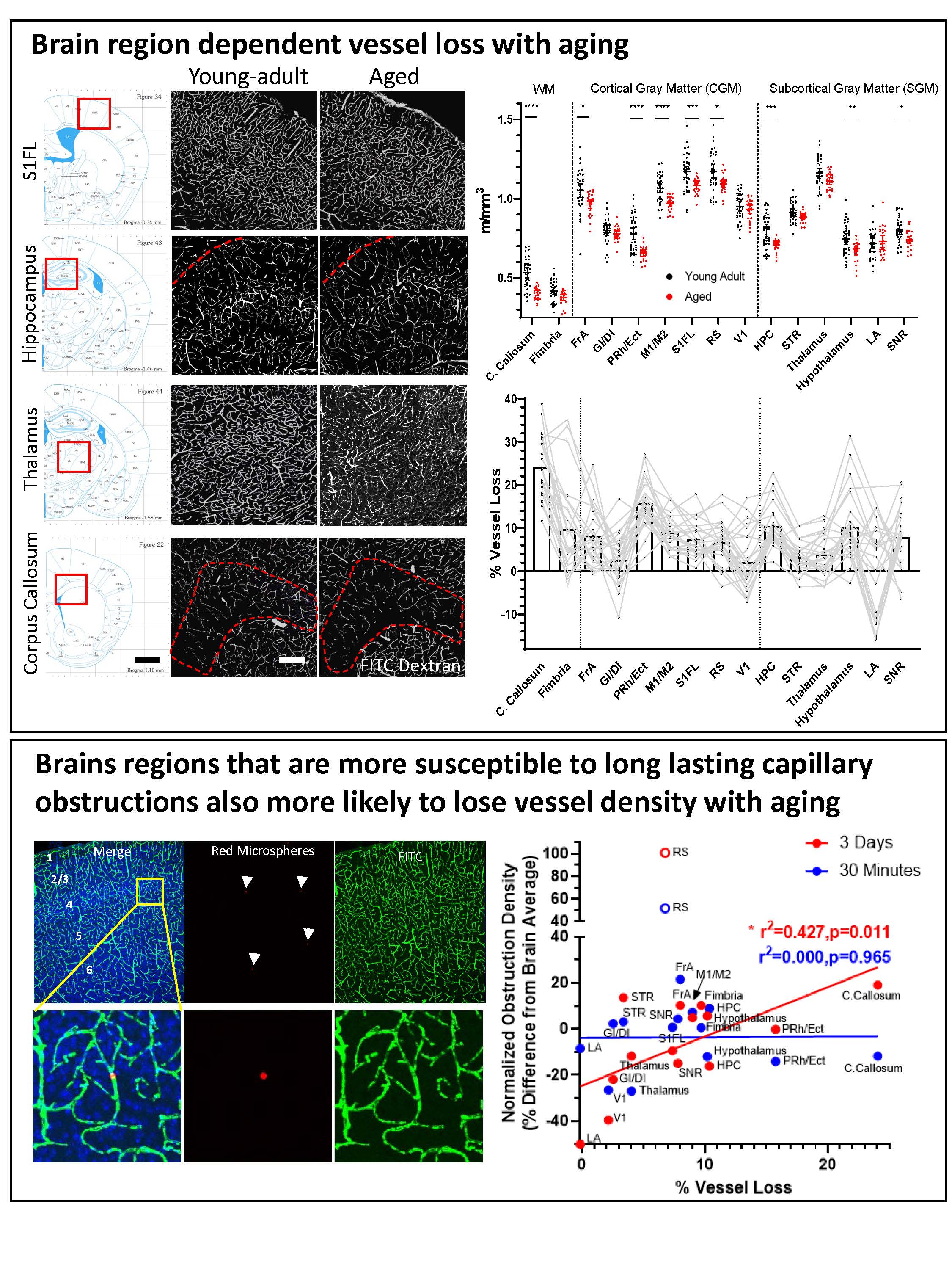
For more information see: Reeson, P, Choi K and Brown CE. 2018. VEGF signaling regulates the fate of obstructed capillaries in mouse cortex. eLIFE.
https://doi.org/10.7554/eLife.33670
For more information see: Tennant KA, Taylor S, White E, Brown CE. 2017. Optogenetic rewiring of thalamocortical circuits to restore function in the stroke injured brain. Nature Communications Jun 23;8:15879. doi: 10.1038/ncomms15879.
Focal ischemic stroke leads to a long-lasting increase in vascular permeability in the peri-infarct cortex of young adult and aged mice. 1. Whole-brain image showing Evans blue dye extravasation around the cerebral infarct 3 h after initiating photothrombosis. 2. Representative confocal image showing the halo of Evans blue fluorescence around the cerebral infarct. This halo of diffuse fluorescence usually extends 300–500 micrometers from the infarct border, which we refer to as ‘‘peri-infarct’’ (PI) cortex. Note that cortical areas more distant to the infarct exhibit little to no fluorescence. 3. A toluidine blue stained section showing the infarct core (above right) and peri-infarct region (below right) 72 h after stroke in an aged mouse. The vessels inside the infarct core were either plugged or unrecognizable. Open perfused microvessels were evident in the peri-infarct zone (below right). Close inspection reveals vacuolated endothelium and swollen astrocyte endfeet. 4. Electron microscopic images of capillaries in sham (left), and in the peri-infarct zone of 3 h (middle) and 72 h post-stroke cortical regions. High magnification views of the details of the endothelium reveal intact tight junctions (black arrows) and an increase in vesicles (arrowheads) within the endothelium. Vacuole formation in the endothelium was typical of capillaries in the peri-infarct zone.
"longitudinal in vivo two-photon images of vascular networks in the peri-infarct cortex of diabetic mice subjected to ischemic stroke. For more details see: Tennant K and Brown CE. 2013. Diabetes augments in vivo microvascular blood flow dynamics after stroke. Journal of Neuroscience 33(49):19194-19204.
"(A) Photomicrograph of distal middle cerebral artery (MCA) through the intact mouse skull. Adjacent image shows skull afer masking procedure to restrict photoactivation to the distal MCA. Inset boxes show the segment of the MCA (labeled with Texas red dextran) that will be photoactivated. (B) Transcranial laser speckle contrast images of cortical blood flow at baseline and during 5, 15, 30 and 60 min of photoactivation. (C) surface vasculature labeled with Evans blue dye 24 hours after sham operation or stroke induction while awake. (D) Infarct volumes for awake or anesthetized mice receiving a glutamate receptor (GluN2B; Ro25-6981) antagonist or alpha-4 nicotinic receptors antagonist (DHβE).
Note the neuroprotective effect of the nicotinic antagonist is only evident in awake mice, perhaps because isoflurane is a known nicotinic receptor blocker. For more details, see Seto A, Taylor S, Trudeau D, Swan I, Leung J, Reeson P, Delaney KR, Brown CE.2014. Induction of ischemic stroke in awake freely moving mice reveals that isoflurane anesthesia can mask the benefits of a neuroprotection therapy.Frontiers in Neuroenergetics Apr 3;6:1.”
"In vivo voltage sensitive dye imaging reveals impaired cortical re-mapping of the forelimb sensory representation 14 weeks after stroke in diabetic and insulin treated mice. A: Montage shows forelimb evoked cortical depolarizations in sham stroke non-diabetic and diabetic mice. B: Montage shows the loss of forelimb-evoked cortical responses in regions next to stroke (indicated by white circle) in both non-diabetic and diabetic mice. After 14 weeks recovery from stroke, cortical responses to the forepaw re-emerge in peri-infarct cortex of non-diabetic. However, this re-mapping is significantly impaired in diabetic mice, as well as diabetic mice that had blood sugar levels controlled after stroke with insulin."
For more details see: Sweetnam D, Holmes A, Tennant K, Zamani A, Walle M, Jones P, Wong C, Brown CE. 2012 Diabetes impairs cortical plasticity and functional recovery following ischemic stroke. Journal of Neuroscience 32(15):5132-5143.
Top Left: In vivo voltage sensitive dye imaging of whisker-evoked depolarizations in the barrel cortex. Note that depolarizations (warm colors) are prominent in the barrel-field and secondary somatosensory cortex (right side of barrel-field). Top right: Low magnification confocal image showing the distribution of GABA-ergic neurons (red) in the barrel-field. Bottom right: High magnification confocal image reveals that YFP labelled alpha-4 nAChRs target GABA-ergic neurons in layer 4.Bottom left: Electron micrograph showing that alpha-4 nAChRs are expressed in neuronal cell bodies in putative peri/extra synaptic regions.
Brown CE, Sweetnam D, Beange M, Nahirney PC, Nashmi R. 2012. Alpha-4 nicotinic acetylcholine receptors modulate experience-based cortical depression in the adult somatosensory cortex. Journal of Neuroscience: 32(4): 1207-1219.

 Roobina Boghozian, Post-doc
Roobina Boghozian, Post-doc

Sorabh Sharma, Post-doc
My primary interest is to study how diabetes alters the neurological function. Recently, increased prevalence of neurodegenerative diseases have been reported in patients suffering from Type 2 diabetes. However, the underlying mechanisms of association between these conditions are still elusive. During my doctorate, I explored the possible involvement of epigenetic modifications in diabetes induced neurodegeneration. We reported that dysregulation of epigenetic pathways leads to cognitive deficit and amyloid-β accumulation in hippocampus of mice. We also examined the therapeutic potential of histone deacetylases inhibitors, during these co-morbid conditions. As a Post doc in the Brown lab, I will be studying how diabetes affects the rates of capillary obstruction and/or pruning. Further, we will focus to elucidate mechanistic understanding of how obstructions are cleared. This could be used in a therapeutic manner to preserve brain micro-circulation and function throughout the lifespan.

Eslam Mehina, Ph.D., Neuroscience Graduate Program
Research Interests: My project aims to decipher the role of microglia in responding to damaged microvessels in the brain, and in contributing to their repair and ultimate fate after hemorrhagic insults. Diabetic patients are at increased risk of suffering from cerebrovascular disease, so I am studying whether microglial dynamics and responses are disturbed in mice with a model of insulin-dependent diabetes. If I find that they are, I will also be investigating means by which these changes can be reversed to prospectively reduce the risk of developing these pathologies.
 Mohammad Motaharinia, M.Sc., Neuroscience Graduate Program
Mohammad Motaharinia, M.Sc., Neuroscience Graduate Program
 Ben Schager, M.Sc., Neuroscience Graduate Program
Julia Templeman, Volunteer/ Honours Student
Ben Schager, M.Sc., Neuroscience Graduate Program
Julia Templeman, Volunteer/ Honours Student
This could be you! We are currently seeking applications for new lab members. Please contact Dr. Brown for details at brownc@uvic.ca.
Kelly Tennant, PhD - Post Doctoral Fellow
September 2011 - January 2018

Patrick Reeson, Ph.D., Neuroscience Graduate Program
Research Interests: Diabetics are more likely to suffer a stroke, and when they do their prognosis for recovery is significantly worse. Previous work in the lab has established that the surviving cortical tissue in the diabetic brain is impaired in its ability to adapt to facilitate recovery. Given the importance of the cerebraovasculature to neuronal function and the widespread vascular pathologies associeated with diabetes, Patrick's work aims to understand how vascular dysfunction in the diabetic brain after stroke impairs plasticity and recovery. Patrick utilizes confocal and 2 photon microscopy to image the cerebrovasculature as well as molecular methods to study key angiogenic and vascular permeability factors such as VEGF.
Graduated June 2018

Kim Gerrow, Ph.D. Post Doctoral Fellow
Research Interests: During stroke recovery, pyramidal neurons in the peri-infarct cortex undergo structural re-arrangements, and in contrast, very little is known about how stroke affects the morphology of cortical interneurons. Many interneurons release GABA and/or peptides that regulate cortical excitability, which is profoundly disrupted after stroke. Of note, interneurons expressing vasoactive intestinal peptide (VIP) specialize in inhibiting other classes of inhibitory cortical neurons, such as those expressing parvalbumin (PV) and somatostatin (SOM). Using longitudinal in vivo imaging and Cre-dependent transgenic mouse lines, we are currently investigating how stroke alters the growth and stability of dendritic arbors, spines and axons in interneurons.
Stephanie Taylor - MSc StudentAngela Seto - MSc Student
June 10, 2013, Angela successfully defended her MSc thesis titled: Role of the a4B2 nicotinic acetylcholine receptor in stroke recovery.
Andrew Sweetnam-Holmes - MSc Student
August 15, 2013, Andrew successfully defended his MSc thesis titled: Diabetes exacerbates the loss of dendritic spines after ischemic stroke.
Dani Sweetnam-Holmes - MSc Student
November 2011, Dani successfully defended her MSc thesis titled "Diabetes impairs cortical plasticity and functional recovery following ischemic stroke". Dani is continuing her education in medicine.